News & Articles
Antigen Testing vs. PCR-based COVID-19 Testing: Pros & Cons
Color
As the COVID-19 pandemic continues, new questions around different types of tests and testing models are arising – and the best choice for any given population isn’t always clear. Antigen testing and rapid point-of-care tests are facing new questions about their accuracy, particularly in populations where preventing pre-symptomatic transmission is critical. Nucleic acid amplification tests (NAAT), including PCR-based strategies like RT-LAMP and RT-PCR, while more sensitive and accurate, can take longer to return results.
At Color, we use RT-LAMP, a PCR-based NAAT test for our large population programs, including San Francisco’s CityTestSF program and USC’s plan to return safely to campus. As testing becomes more available and case data across the country continues to evolve, understanding the difference between antigen testing and NAAT testing can help leaders make decisions about which model works best for their population.
Antigen or Nucleic Acid Amplification Tests?
Diagnostic tests are measured on two different dimensions: specificity and sensitivity. Specificity refers to how well a test can identify someone who is not infected and sensitivity refers to how well a test can identify someone who is infected.
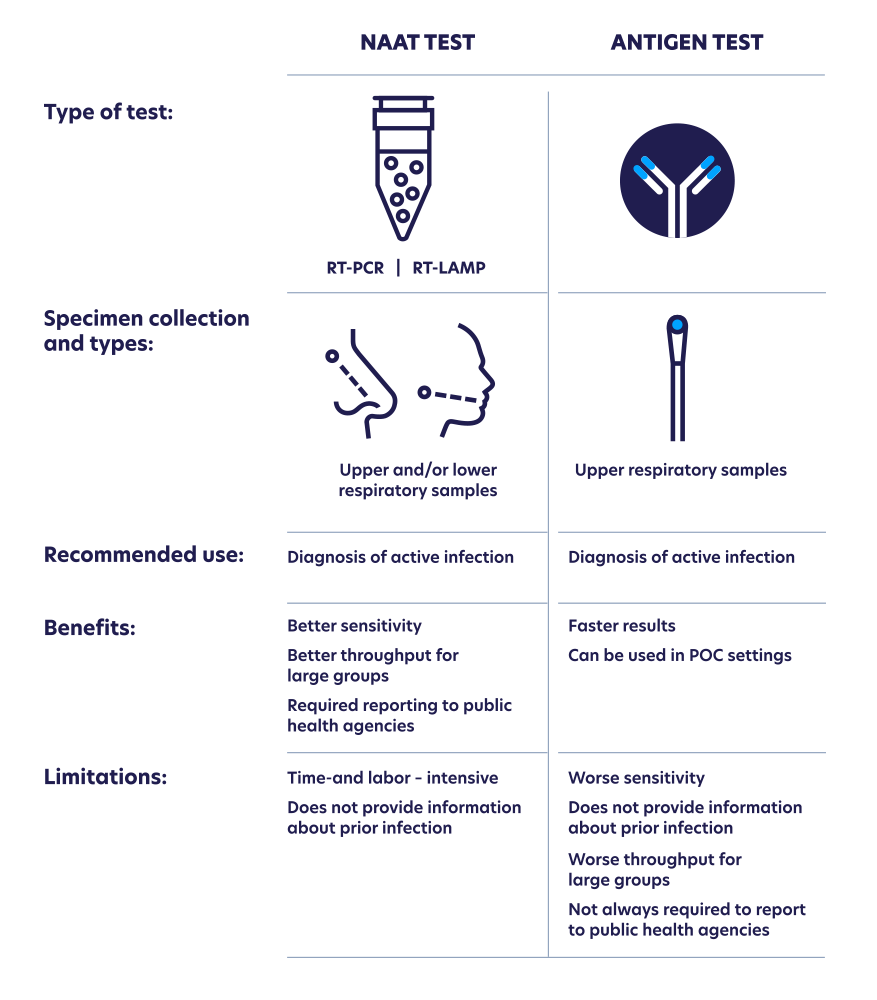
Antigen testing detects the presence of a protein that is part of the SARS-CoV-2 virus, which causes COVID-19. Effectively, antigen testing identifies fragments of viral proteins that can reveal if someone is infected with the virus. However, negative test results from an antigen test do not always rule out infection. According to the FDA, while this type of test is very specific for the virus, it isn’t as sensitive as other options – this means there is a higher chance of false negatives. However, they are often less expensive and can provide an individual result within minutes, making speed and cost some of the main advantages.
NAAT (nucleic acid amplification tests), on the other hand, detect the genetic material from the virus, the RNA, to identify the presence of SARS-CoV-2. NAAT typically use PCR-based strategies, which include an amplification step that makes it possible to identify very small quantities of the virus’ RNA – this makes them more sensitive than antigen testing, leading to fewer false negatives. NAAT tests, however, are typically more expensive to run and take longer than antigen tests. Instead of returning an individual result within 15-30 minutes, results are typically returned within 24-72 hours.
Which test is the best option?
When it comes to selecting the best test for managing the spread of COVID-19, decision makers at organizations responsible for the health of large populations are having to balance accuracy with speed and throughput.
While quicker turn-around time is ideal, returning results in under 48 hours has still been shown to be effective in containing larger community outbreaks. That evidence, coupled with the greater sensitivity, make NAAT testing a good option for large scale testing. We modeled this type of scenario with Dr. Carl Bergstrom from the University of Washington and found that when results can be delivered 1-2 days after sample collection, public health experts are still able to intervene quickly enough to prevent or dramatically mitigate an outbreak event in most settings.
While the speed of antigen testing is useful in certain settings, building the logistics for rapid testing at scale has proven to be difficult and expensive. This type of test is fast for a handful of people, but this speed is almost impossible to achieve for thousands of people, unless an organization has access to thousands of point-of-care machines. Although it takes approximately 15 minutes to run a single antigen test, about 100 NAAT can be run in just an hour and a half. This makes NAAT a more useful approach for situations in which large groups need to be tested at once. For example, if every passenger boarding an international flight was required to be antigen tested on the day of travel, the lines and wait times for a test would be long. Our partnership with United allows pre-flight testing to be spread out over 72 hours prior to flying – and passengers can still select rapid point-of-care testing from another partner, if that works better for their family and travel plans.
Using large-scale antigen testing also has concerning public health consequences. Since NAAT are run by labs required to report positive results to state departments of public health, they allow for more consistent outbreak mitigation and more rapid contact tracing efforts. If an individual receives a positive result on an antigen test, that individual has the responsibility to report the result and contact trace, which isn’t always the best way to keep populations safe. For example, a person who wants to continue working or travel may choose to hide their positive result, if it means they would have to take sick time or cancel their trip. With NAAT results, hiding a positive result is much more difficult.
Finally, there are populations where preventing pre-symptomatic transmission is critical, like nursing homes. Antigen testing may not be the answer in these contexts; some experts have expressed concern with the results of antigen testing in congregate settings.
What’s next?
When it comes to bringing our country safely back to normal, there are certainly a number of COVID-19 testing strategies that can be implemented to manage outbreaks. Antigen testing can play an important role in a comprehensive testing strategy, but unfortunately, relying solely on antigen and rapid testing has not proven to be a reliable testing method to prevent outbreaks. Outbreaks among populations who have relied only on this type of testing have received more attention in recent days.
At Color, we have deep experience in delivering fast, efficient, and accurate results to large populations. We’ve consistently seen NAAT be a stronger option for large populations. Our priority is bolstering the public health response to this pandemic and at Color, we are constantly examining what approaches can work best for our partners, as testing technology can evolve rapidly. Our work this year has illustrated that utilizing the strength of NAAT is helping leaders and experts keep communities safe.
About Color
Color is a leader in distributed healthcare and clinical testing. Color makes population-scale healthcare programs accessible, convenient, and cost-effective for everyone. Color works with health systems, employers, and national health initiatives around the world including the million-person All of Us Research Program by the National Institutes of Health. For more information about Color and its response to COVID-19, visit www.color.com.



