Take control
of cancer.
Change the story of cancer in your population with Color’s comprehensive cancer solution, designed in partnership with the American Cancer Society.
Color is the only solution that combines early detection and comprehensive cancer management before, during, and after a diagnosis.

We’ve partnered with the American Cancer Society to make cancer detection and management more convenient, more compassionate, and more comprehensive for your employees and members.
Together, we’re working to end cancer as we know it.
We drive impact by, first, helping detect cancer earlier and, second, by managing cancer care more effectively.
Meaningful engagement
>20%
average member participation in the first year
Demonstrated health outcomes
77%
average increase in screening adherence
Exceptional care
4.95/5
average patient satisfaction score
Compounding savings
~$2M
in projected savings for a population of 10K individuals
Detecting cancer early makes the single biggest impact on survival rates and healthcare costs.
- Adapted from McGarvey N, Gitlin M, Fadli E, Chung KC. Increased healthcare costs by later stage cancer diagnosis. BMC Health Serv
- SEER*Explorer Application , National Cancer Institute
Our approach is evidence-based, proactive, and comprehensive—without the friction of traditional care.
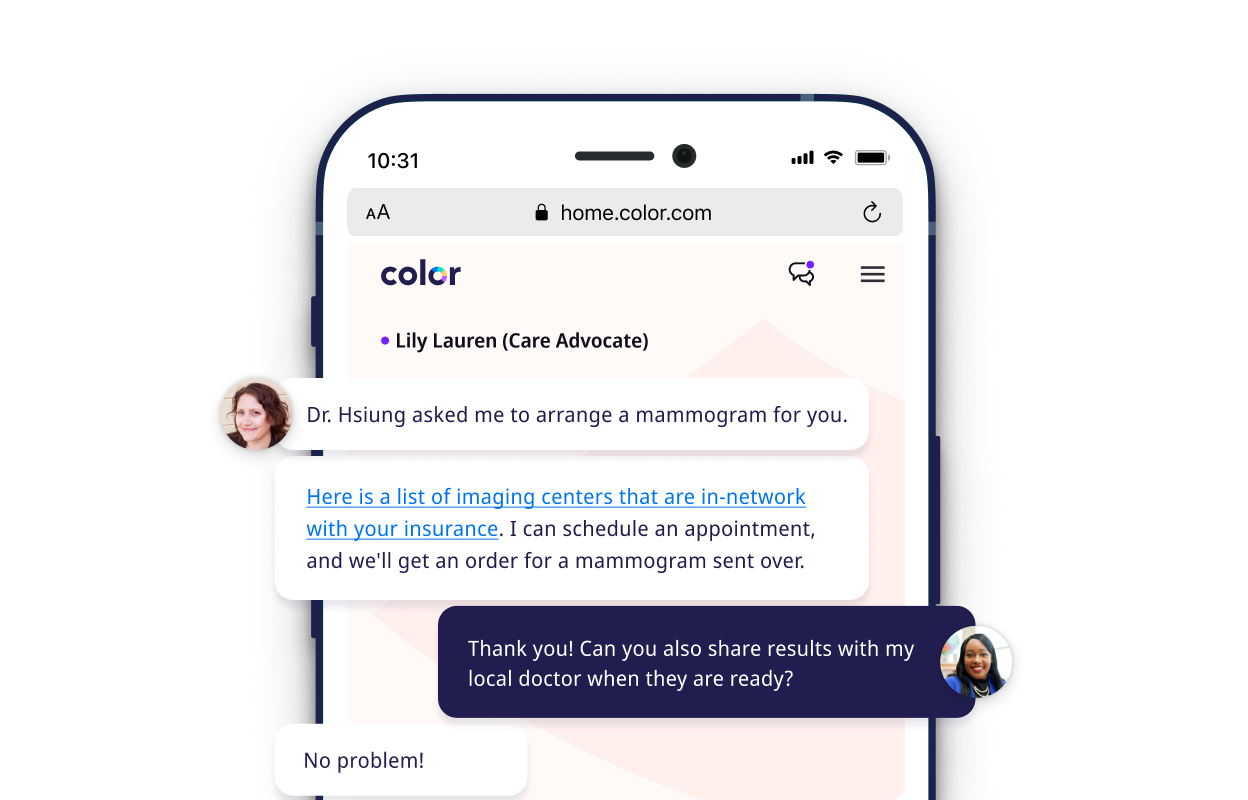
We provide better integrated care.
Our care advocacy teams, nationwide medical group, and integrated mental health support groups help individuals get on track with the right screenings, provide the best care from follow-up to diagnosis, and take on the logistical burden of in-person care, such as scheduling and referrals.

We shorten time to diagnosis and route your population to care faster.
We help detect cancer earlier by making screening more accessible and convenient, at home or in person, and by shortening time from screening to diagnosis.
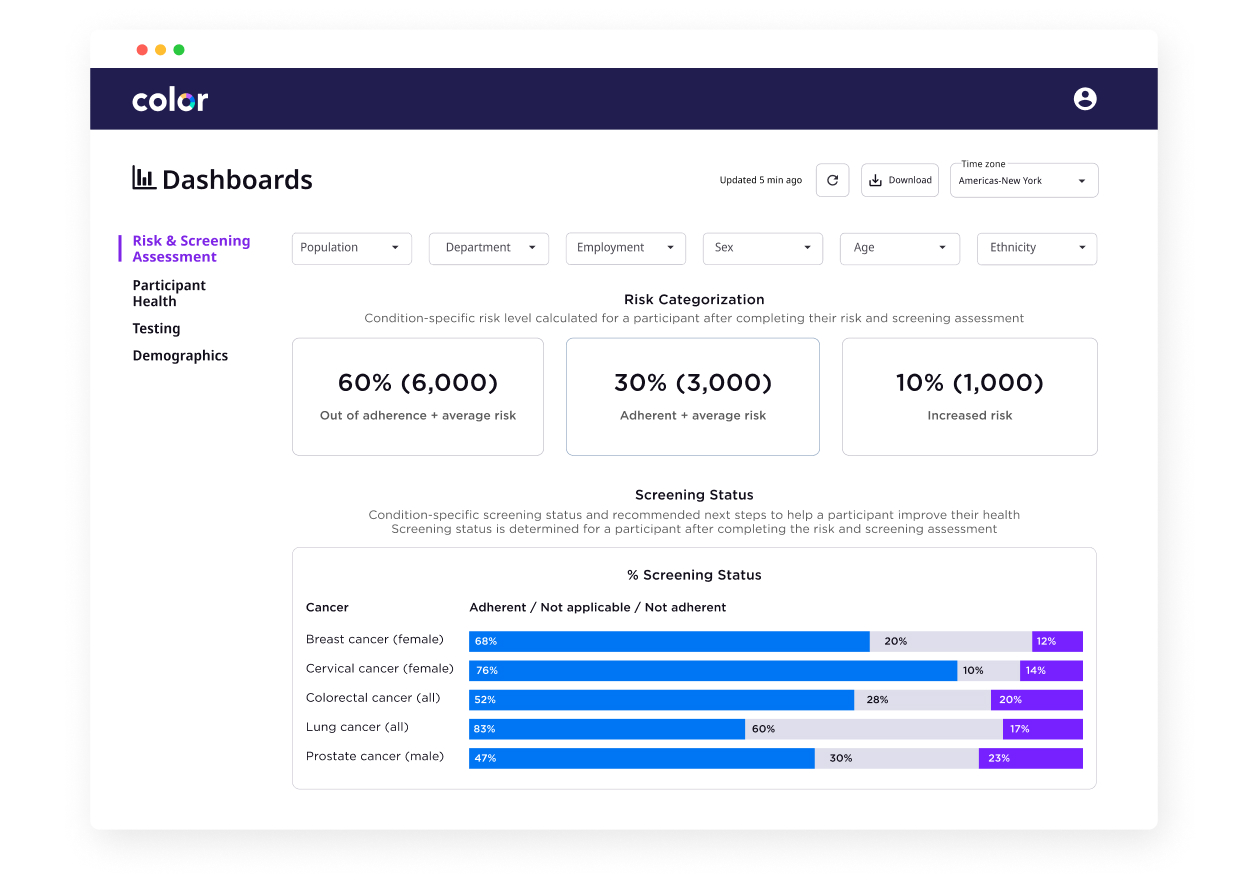
We give you visibility into cancer risk within your population.
We understand each individual’s unique risk for cancer and provide real time visibility on screening gaps and high-risk groups within your population.

We educate and engage your population.
We’ve developed onsite, virtual, and digital educational programming, covering topics such as self-examination, genetic risk, and preventive behaviors, to continuously engage your population in their health.
We increase survival rates and reduce cancer-related costs for:
Designed for people. Built for populations.

Color’s mission is to help everyone lead the healthiest lives science and medicine allow. To do so, we deliver integrated care that removes the logistical, technological, financial, and cultural barriers that accompany traditional healthcare delivery.

With nearly a decade of experience in the cancer and high-risk space, Color has served millions and partnered with innovators such as the National Institutes of Health, the International Brotherhood of Teamsters, and Salesforce.

Our approach is both targeted and scalable—built with infrastructure that meets large organization’s needs, while remaining designed for accessible, patient-centered care.
Speak with an expert.
Want to get in touch with our team? We’re just a click away.
New to Color?
Whether you want to learn more about Color or you’re ready to start your journey with us, you’re in the right place.
Already a customer?
If you need help with your account, sample submission, or other service-related issue, we’re here.
New to Color?
Whether you want to learn more about Color or you’re ready to start your journey with us, you’re in the right place.
Already a customer? Get support.
Call us toll-free between 6am and 5pm PT at (844) 352-6567. Those outside the US can reach us at +1 650-651-7116. We’re also available via email at support@color.com.
For frequently asked questions please visit support.color.com.




