Color becomes the first and only Virtual Cancer Clinic to achieve ASCO certification.
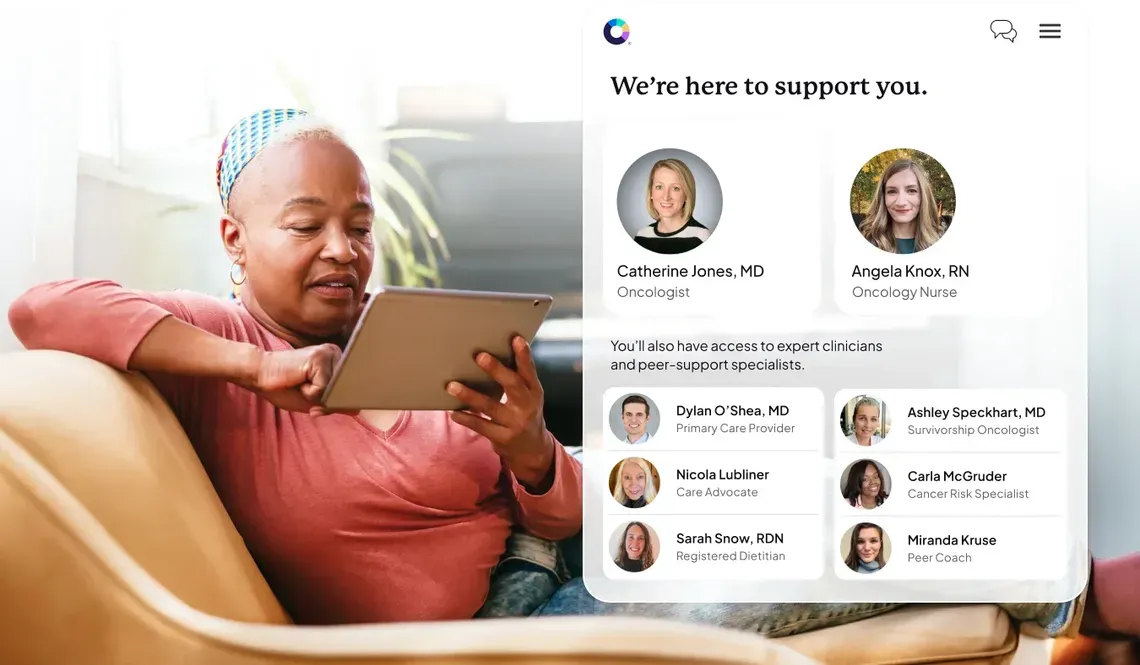
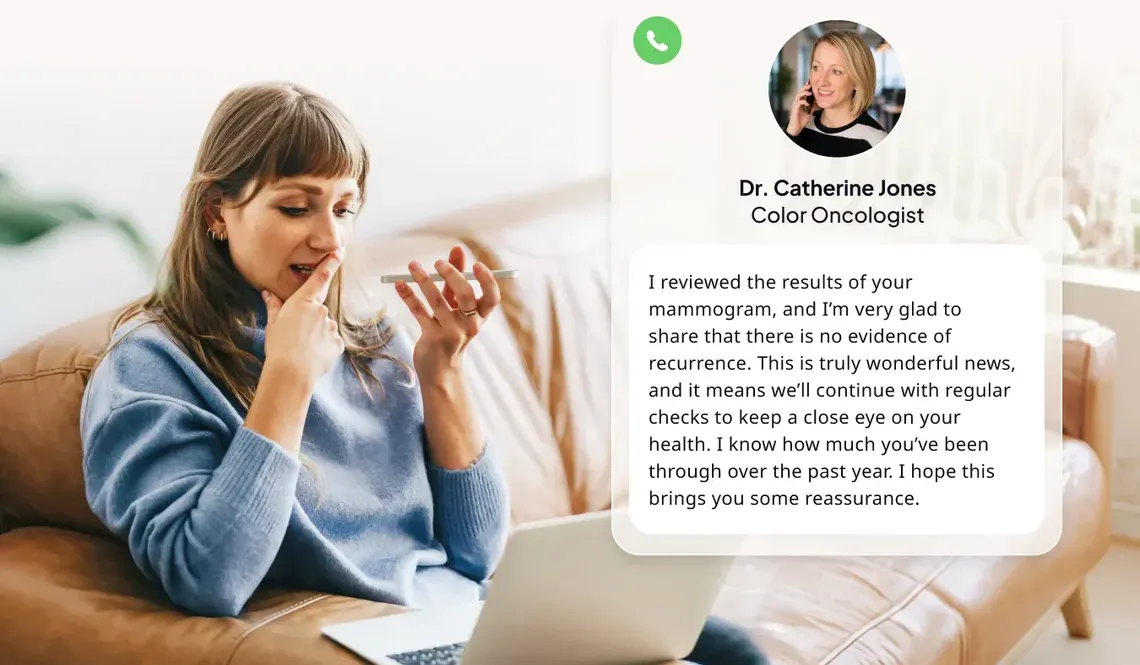
True employee engagement, clinical outcomes, and cost control across the entire cancer care journey is the reality, with Color







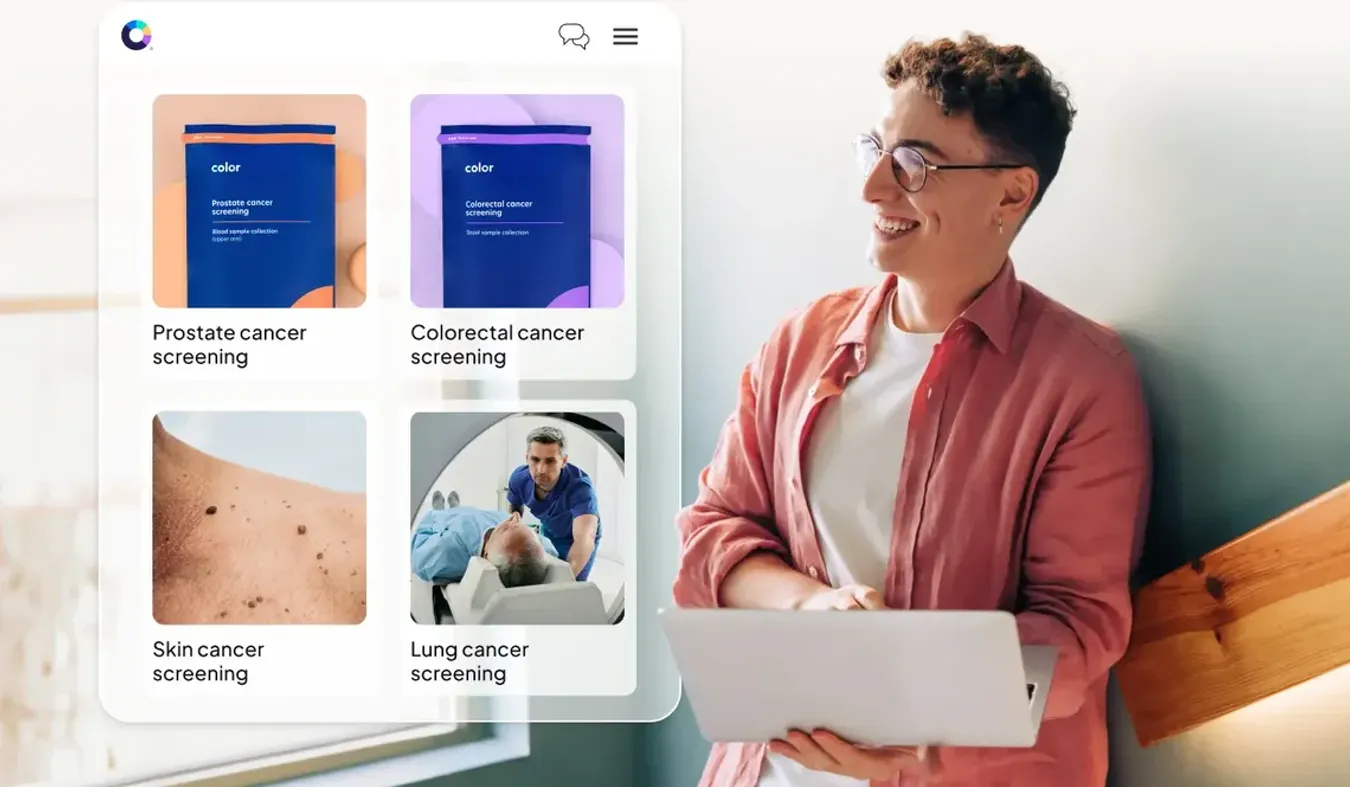
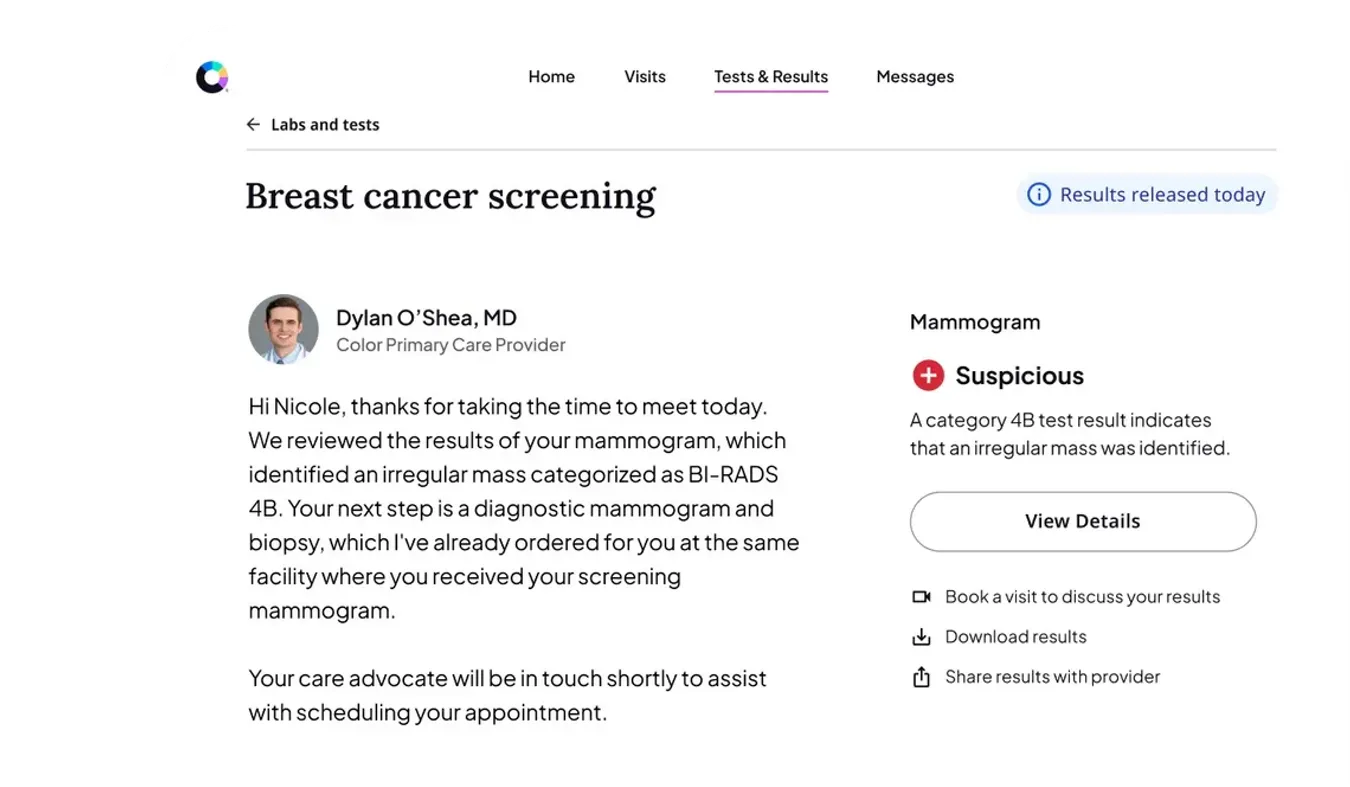
With cancer as the largest driver of employer healthcare spend, you need to look for answers beyond uncontrollable drug prices. Instead, focus on avoidable issues like low screenings, lack of followup, delayed diagnoses, uncoordinated care, and limited treatment management through survivorship. This is where you can truly impact cost drivers and achieve better outcomes. Most traditional programs are reactive, showing up too late to improve outcomes and costs.
With Color on your side, you can change the trajectory for good.

faster diagnosis
increase in screening adherence
of costly care gaps closed
saved per patient
saved per survivor
in Year 1